 3D VSEPR app helps you to visualize the shapes of the. Kit includes 1 black carbon tetrahedral 9mm, 9 white hydrogen monovalent 9mm, 1 black carbon linear 9mm, 1 beige trigonal 9mm, 1 yellow sulfur octahedral 9mm, 6 light green fluorine monovalent 9mm, 1 purple phosphorus trigonal bipyramidal 9mm, 5 dark green chlorine monovalent 9mm, 1 yellow sulfur divalent 9mm, 2 red oxygen monovalent 9mm, 22 - 20mm grey straw bond. It is impossible to learn about the shapes of VSEPR models in a 2D page because these arent in 2D. In this lesson we are going to go over a couple of fundamental topics important for any organic chemistry student. Build the following molecules: Linear CO2, Bent SO2, Trigonal Planar BH3, Tetrahedral CH4, Trigonal bipyramidal PCl5, Octahedral SF6. Organic Chemistry VSEPR Theory and Hybridization. VSEPR Theory is short for Valence Shell Electron Pair Repulsion Theory, a method of organizing molecules based on their geometric structures.  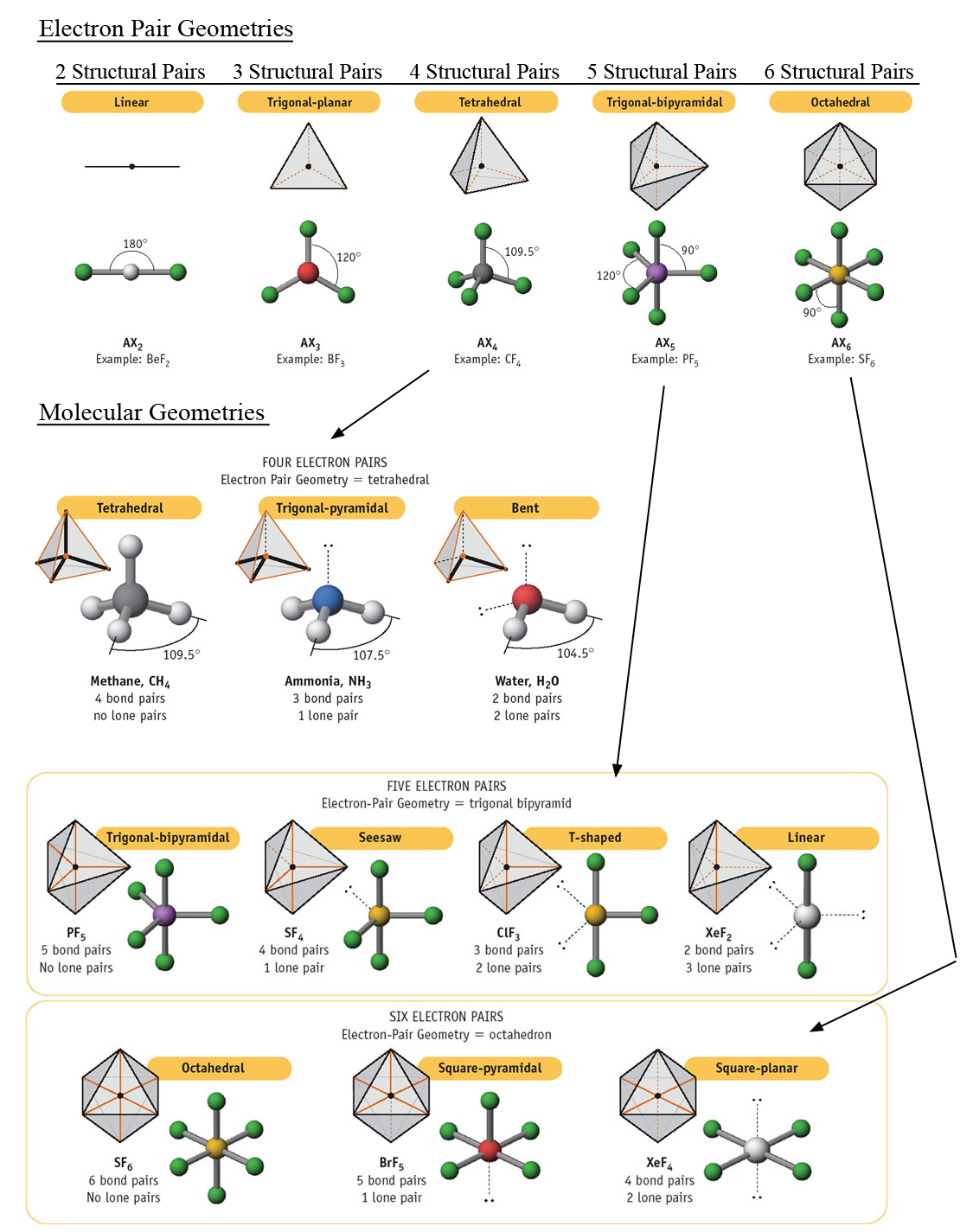
Molecular Models 50 Piece Basic VSEPR Theory Molecule Shapes Models Kitīuild the following molecules Linear CO2, Bent SO2, Trigonal Planar BH3, Tetrahedral CH4, Trigonal bipyramidal PCl5, Octahedral SF6 | Build examples of the six basic VSEPR theory shapes with this economical and easy to assemble kit | Kit includes 1 black carbon tetrahedral 9mm, 9 white hydrogen monovalent 9mm, 1 black carbon linear 9mm, 1 beige trigonal 9mm, 1 yellow sulfur octahedral 9mm, 6 light green fluorine monovalent 9mm | Also includes 1 purple phosphorus trigonal bipyramidal 9mm, 5 dark green chlorine monovalent 9mm, 1 yellow sulfur divalent 9mm, 2 red oxygen monovalent 9mm, 22 - 20mm grey straw bondīuild examples of the six basic VSEPR theory shapes with this economical and easy to assemble kit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
